|
|
|
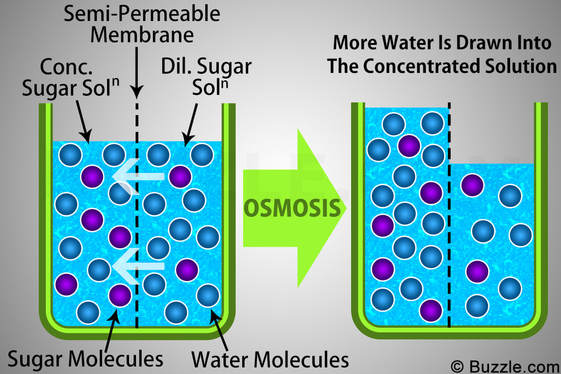
Osmosis is the net movement of water molecules across a semi-permeable membrane from a region of low solute concentration to a region of high solute concentration (until equilibrium is reached)
- Water is considered the universal solvent – it will associate with, and dissolve, polar or charged molecules (solutes)
- Because solutes cannot cross a cell membrane unaided, water will move to equalise the two solutions
- At a higher solute concentration there are less free water molecules in solution as water is associated with the solute
- Osmosis is essentially the diffusion of free water molecules and hence occurs from regions of low solute concentration
Skill:
1.4.S.1: Estimation of osmolarity in tissues by bathing samples in hypotonic and hypertonic solutions. PRESCRIBED PRACTICAL 2
Objectives:
1.4.S.1: Estimation of osmolarity in tissues by bathing samples in hypotonic and hypertonic solutions. PRESCRIBED PRACTICAL 2
Objectives:
- Define osmolarity, isotonic, hypotonic and hypertonic.
- Calculate the percentage change between measurement values.
- Calculate the mean value of a data set.
- Calculate the standard deviation value of a data set.
- State that the term standard deviation is used to summarize the spread of values around the mean, and that 68% of the values fall within one standard deviation of the mean.
- Explain how the standard deviation is useful for comparing the means and the spread of data between two or more samples.
- Determine the correct type of graph to represent experimental results.
- State that error bars are a graphical representation of the variability of data.
- Accurately graph mean and standard deviation of data sets.
- Determine osmolarity of a sample given changes in mass when placed in solutions of various tonicities.
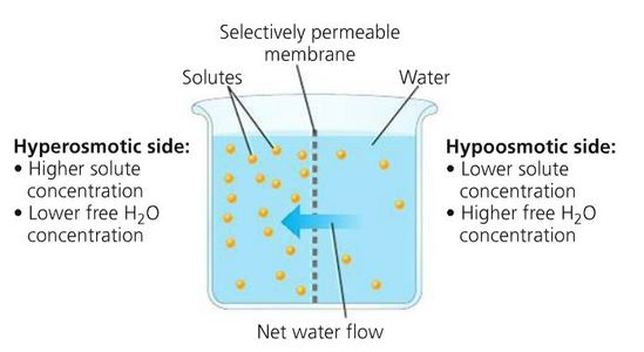
Osmolarity is a measure of solute concentration, as defined by the number of osmoles of a solute per litre of solution (osmol/L). Solutions may be loosely categorised as hypertonic, hypotonic or isotonic according to their relative osmolarity
- Solutions with a relatively higher osmolarity are categorised as hypertonic (high solute concentration ⇒ gains water)
- Solutions with a relatively lower osmolarity are categorised as hypotonic (low solute concentration ⇒ loses water)
- Solutions that have the same osmolarity are categorised as isotonic (same solute concentration ⇒ no net water flow)
Osmotic Movement between Solutions: Hypertonic (left) and Hypotonic (right
Estimating Osmolarity
The osmolarity of a tissue may be interpolated by bathing the sample in solutions with known osmolarities
The osmolarity of a tissue may be interpolated by bathing the sample in solutions with known osmolarities
- The tissue will lose water when placed in hypertonic solutions and gain water when placed in hypotonic solutions
- Water loss or gain may be determined by weighing the sample before and after bathing in solution
- Tissue osmolarity may be inferred by identifying the concentration of solution at which there is no weight change (i.e. isotonic)
Application:
1.4.A.2: Tissues or organs to be used in medical procedures must be bathed in a solution with the same osmolarity as the cytoplasm to prevent osmosis
Objectives:
1.4.A.2: Tissues or organs to be used in medical procedures must be bathed in a solution with the same osmolarity as the cytoplasm to prevent osmosis
Objectives:
- Explain what happens to cells when placed in solutions of the same osmolarity, higher osmolarity and lower osmolarity.
- Outline the use of normal saline in medical procedures.
Tissues or organs to be used in medical procedures must be kept in solution to prevent cellular dessication. This solution must share the same osmolarity as the tissue / organ (i.e. isotonic) in order to prevent osmosis from occurring.
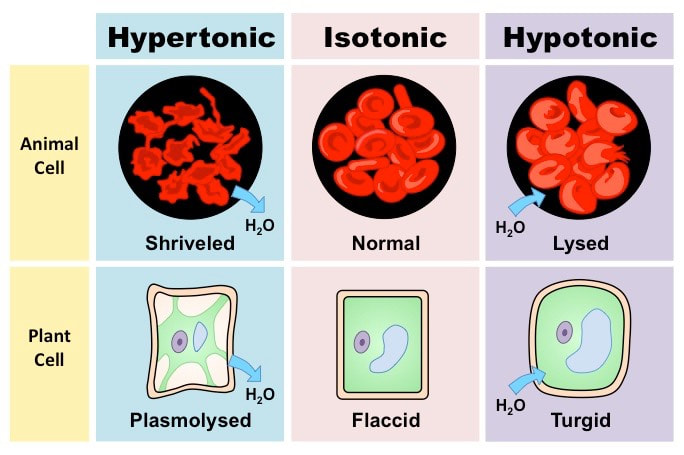
Uncontrolled osmosis will have negative effects with regards to cell viability:
Uncontrolled osmosis will have negative effects with regards to cell viability:
- In hypertonic solutions, water will leave the cell causing it to shrivel (crenation)
- In hypotonic solutions, water will enter the cell causing it to swell and potentially burst (lysis)
- In hypertonic solutions, the cytoplasm will shrink (plasmolysis) but the cell wall will maintain a structured shape
- In hypotonic solutions, the cytoplasm will expand but be unable to rupture within the constraints of the cell wall (turgor)
Summary of the Effects of Solute Concentrations on Cells