electron configuration
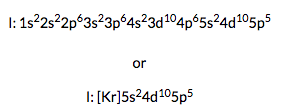
The electron configuration is the standard notation used to describe the electronic structure of an atom. When assigning electrons to orbitals, we must follow a set of three rules: the Aufbau Principle, the Pauli-Exclusion Principle, and Hund's Rule.
To help describe the appropriate notation for electron configuration, it is best to do so through example. For this example, we will use the iodine atom. There are two ways in which electron configuration can be written as depicted to the right:
In both of these types of notations, the order of the energy levels must be written by increased energy, showing the number of electrons in each subshell as an exponent. In the short notation, you place brackets around the preceding noble gas element followed by the valence shell electron configuration. The periodic table shows that kyrpton (Kr) is the previous noble gas listed before iodine. The noble gas configuration encompases the energy states lower than the valence shell electrons.
To help describe the appropriate notation for electron configuration, it is best to do so through example. For this example, we will use the iodine atom. There are two ways in which electron configuration can be written as depicted to the right:
In both of these types of notations, the order of the energy levels must be written by increased energy, showing the number of electrons in each subshell as an exponent. In the short notation, you place brackets around the preceding noble gas element followed by the valence shell electron configuration. The periodic table shows that kyrpton (Kr) is the previous noble gas listed before iodine. The noble gas configuration encompases the energy states lower than the valence shell electrons.
rules for writing e configuration
There are a set of general rules that are used to figure out the electron configuration of an atomic species: Aufbau's Principle, Hund's Rule and the Pauli-Exclusion Principle. Before continuing, it's important to understand that each orbital can be occupied by two electrons of opposite spin (which will be further discussed later). The following table shows the possible number of electrons that can occupy each orbital in a given subshell.
Aufbau Principle
The word 'Aufbau' is German for 'building up'. The Aufbau principle, also called the building-up principle, states that electron's occupy orbitals in order of increasing energy. The order of occupation is as follows:
Aufbau Principle
The word 'Aufbau' is German for 'building up'. The Aufbau principle, also called the building-up principle, states that electron's occupy orbitals in order of increasing energy. The order of occupation is as follows:
|
This order of occupation roughly represents the increasing energy level of the orbitals. Hence, electrons occupy the orbitals in such a way that the energy is kept at a minimum. That is, the 7s, 5f, 6d, 7p subshells will not be filled with electrons unless the lower energy orbitals, 1s to 6p, are already fully occupied. Also, it is important to note that although the energy of the 3d orbital has been mathematically shown to be lower than that of the 4s orbital, electrons occupy the 4s orbital first before the 3d orbital. This observation can be ascribed to the fact that 3d electrons are more likely to be found closer to the nucleus; hence, they repel each other more strongly. Nonetheless, remembering the order of orbital energies, and hence assigning electrons to orbitals, can become rather easy when related to the periodic table.
|
Hund's Rule
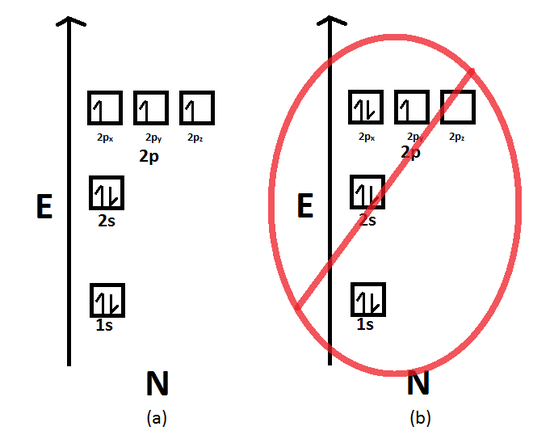
Hund's Rule states that when electrons occupy degenerate orbitals (i.e. same n and l quantum numbers), they must first occupy the empty orbitals before double occupying them. Furthermore, the most stable configuration results when the spins are parallel (i.e. all alpha electrons or all beta electrons). Nitrogen, for example, has 3 electrons occupying the 2p orbital. According to Hund's Rule, they must first occupy each of the three degenerate p orbitals, namely the 2px orbital, 2py orbital, and the 2pz orbital, and with parallel spins. The configuration below is incorrect because the third electron occupies does not occupy the empty 2pz orbital. Instead, it occupies the half-filled 2px orbital. This, therefore, is a violation of Hund's Rule.
Hund's Rule states that when electrons occupy degenerate orbitals (i.e. same n and l quantum numbers), they must first occupy the empty orbitals before double occupying them. Furthermore, the most stable configuration results when the spins are parallel (i.e. all alpha electrons or all beta electrons). Nitrogen, for example, has 3 electrons occupying the 2p orbital. According to Hund's Rule, they must first occupy each of the three degenerate p orbitals, namely the 2px orbital, 2py orbital, and the 2pz orbital, and with parallel spins. The configuration below is incorrect because the third electron occupies does not occupy the empty 2pz orbital. Instead, it occupies the half-filled 2px orbital. This, therefore, is a violation of Hund's Rule.
|
Pauli Exclusion Principle
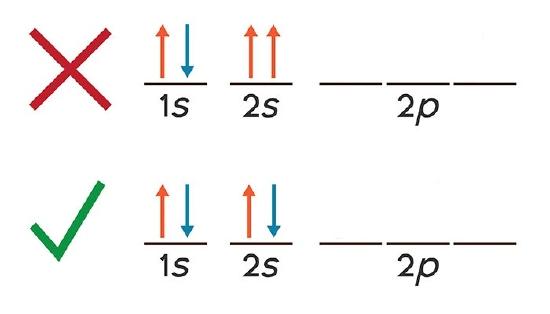
The Pauli Exclusion Principle = An atomic orbital may describe at most two electrons, at least one for that orbital to exist. To occupy the same orbital, two electrons must have opposite spins. If two electrons occupy the same orbital they are said to be paired. Orbitals with one electron are unpaired. |