Understanding:
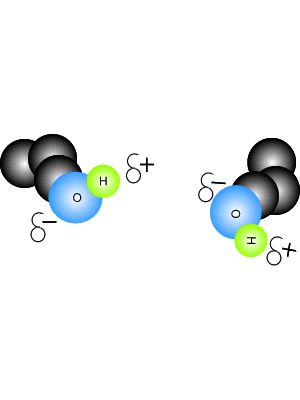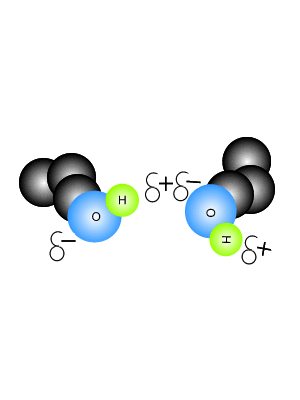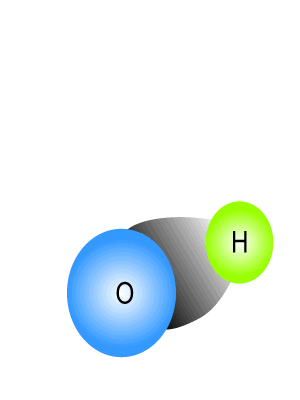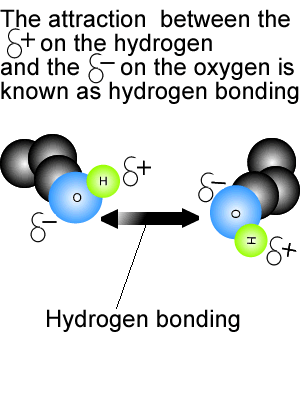
2.2 U.2 Hydrogen bonding and dipolarity explain the cohesive, adhesive, thermal and solvent properties of water.
2.2 U.2 Hydrogen bonding and dipolarity explain the cohesive, adhesive, thermal and solvent properties of water.
Objectives:
[Students should know at least one example of a benefit to living organisms of each property of water. Transparency of water and maximum density at 4°C do not need to be included.]
- Contrast adhesion with cohesion.
- Outline an example of the cohesive property of water being of benefit to life.
- Outline an example of the adhesive property of water being of benefit to life.
[Students should know at least one example of a benefit to living organisms of each property of water. Transparency of water and maximum density at 4°C do not need to be included.]