properties of matter

A property describes how an object looks, feels, or acts. All matter has general properties such as mass, volume, density, color, flammability, etc. There are two main categories of properties: physical and chemical.
Physical Properties: Properties that we observe without changing the identity of the substance. Some examples are:
Physical Properties: Properties that we observe without changing the identity of the substance. Some examples are:
- Feel (Mass, Volume, Texture), Sight (Color, Shape), Sound, Smell, Taste
- Boiling pt., Freezing pt., Melting Pt.
- Conductivity
- Ductility
- Hardness
- Solubility
- Malleability
- Luster
- Density
Chemical Properties: Properties that we observe only when the identity changes. Some examples are:
- Reactivity (reacts with air, water, oxygen, etc.)
- Flammability
- Rusts
- Combustion
- Radioactivity
Unit 2 physical & chemical changes
Skill:
2.U3: Matter can undergo physical and chemical changes
Objective:
* An asterisk indicates objectives covered by the Honors Chemistry class
2.U3: Matter can undergo physical and chemical changes
Objective:
- Define physical change and list several common physical changes
- Define chemical change and list several indications that a chemical change has taken place
- Compare and contrast physical and chemical changes in matter.
- Distinguish between endothermic and exothermic chemical and physical changes.
- Draw and label an energy change diagram for an endothermic and exothermic reaction *
- State the Law of Conservation of Mass and Energy and apply it to chemical changes.
* An asterisk indicates objectives covered by the Honors Chemistry class
physical & chemical change

Matter is constantly changing through natural and artificial means. There are two types of changes: Physical and Chemical Changes.
Physical Change: Any change in size, shape, form, or state of matter.
Physical Change: Any change in size, shape, form, or state of matter.
- All state changes are physical changes
- The identity of the matter stays the same only some physical properties change.
- Most physical changes are reversible.
- Some examples are change in size, shape, state of matter, etc.

Chemical Change: When one type of matter changes into another type of matter with totally different properties.
- Chemical change is the same as a chemical reaction.
- Whenever something burns, a chemical change is taking place.
- Most chemical changes are irreversible because totally new substances are formed.
- Some examples are burning, rusting, combusting, etc.
- Release or absorption of energy in the form of light, heat, or sound.
- A formation of a gas after a chemical reaction.
- A formation of a precipitate (solid) after a chemical reaction.
- Often, a change in color.
ENdothermic & Exothermic Reactions
In endothermic reactions, energy enters. In exothermic reactions, energy exits.
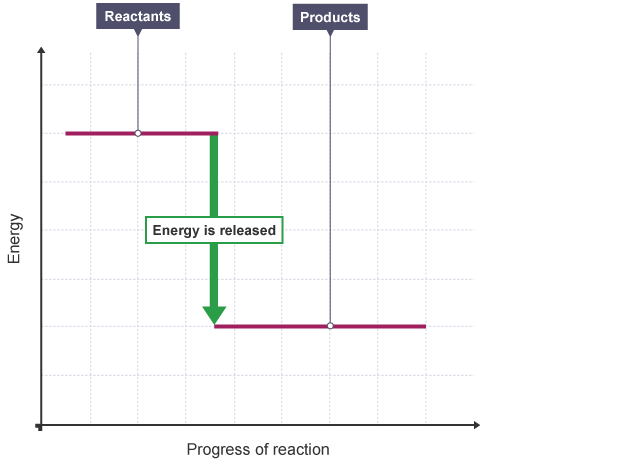
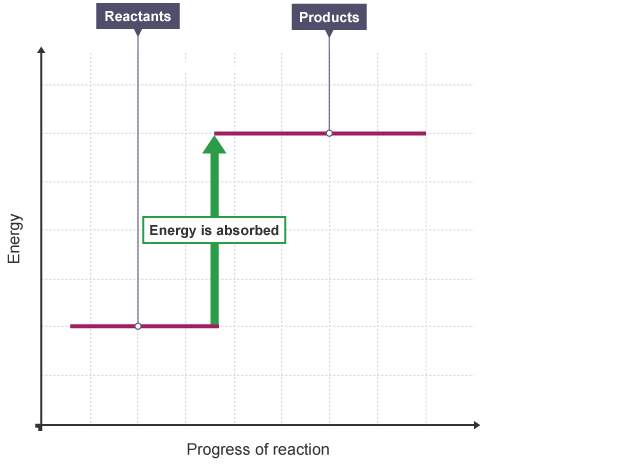
An energy level diagram shows whether a reaction is exothermic or endothermic. It shows the energy in the reactants and products, and the difference in energy between them.
Exothermic reaction
The energy level decreases in an exothermic reaction. This is because energy is given out to the surroundings.
An energy level diagram shows whether a reaction is exothermic or endothermic. It shows the energy in the reactants and products, and the difference in energy between them.
Exothermic reaction
The energy level decreases in an exothermic reaction. This is because energy is given out to the surroundings.
Endothermic reaction
The energy level increases in an endothermic reaction. This is because energy is taken in from the surroundings.
The energy level increases in an endothermic reaction. This is because energy is taken in from the surroundings.