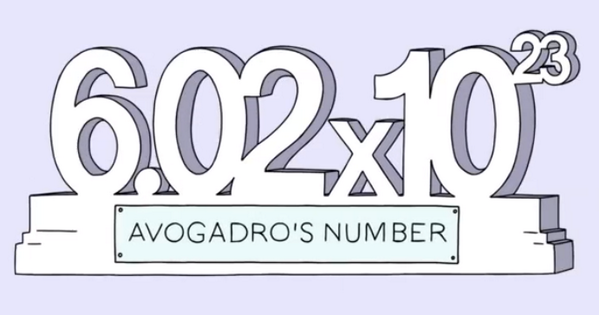Understandings:
7.U1: The mole is a number representing a large quantity.
7.U2: A molecular formula is a whole number multiple of an empirical formula.
Skills:
7.S1: Calculations of mole conversions in chemical reactions.
7.S2: Calculation of mole ratios in a balanced chemical equation.
7.S3: Calculations of percent composition of the elements in a compound and its relation to its empirical and molecular formula
7.S4: Calculations of limiting and excess reagent.
7.S5: Calculations of percent yield using theoretical and actual yield.
7.U1: The mole is a number representing a large quantity.
7.U2: A molecular formula is a whole number multiple of an empirical formula.
Skills:
7.S1: Calculations of mole conversions in chemical reactions.
7.S2: Calculation of mole ratios in a balanced chemical equation.
7.S3: Calculations of percent composition of the elements in a compound and its relation to its empirical and molecular formula
7.S4: Calculations of limiting and excess reagent.
7.S5: Calculations of percent yield using theoretical and actual yield.