ATOMIC STRUCTURE
Atoms are the basis of chemistry. They are the basis for everything in the Universe. You should start by remembering that matter is composed of atoms. Atoms and the study of atoms are a world unto themselves.
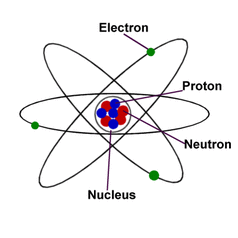
Atoms are the basic units of matter and the defining structure of elements. Atoms are made up of three particles: protons, neutrons and electrons.
Atoms are the basic units of matter and the defining structure of elements. Atoms are made up of three particles: protons, neutrons and electrons.
Basic Parts of the Atom - Protons, Neutrons, Electrons, Nucleus
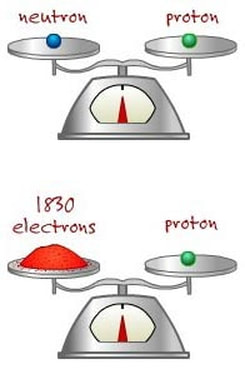
The relative masses of the subatomic particles
The mass of a proton is nearly the same as the mass of a neutron but... the mass of proton (or neutron) is about 2000 times LARGER than the mass of an ELECTRON!!!
The NUCLEUS
The nucleus of the atom is composed of proton and neutron particles. It is understood that "opposite charges" attract and "like-charges" repel. Because the nucleus contains only "like-charged" protons there must exist a force of attraction to keep the positively charged protons together in the nucleus. Chemists predict there exists a "Nuclear Force" that works over extremely small distances (less distance than the width of an atom). When an atomic nucleus grows to a size greater than 83 (with exceptions) the nucleus will break apart naturally giving off what we call radiation. This radiation is exhibited in several forms one being "alpha-particles" and can potentially change the element (by changing the number of protons in the nucleus) in what is known as half-life.