tRANSITIONS
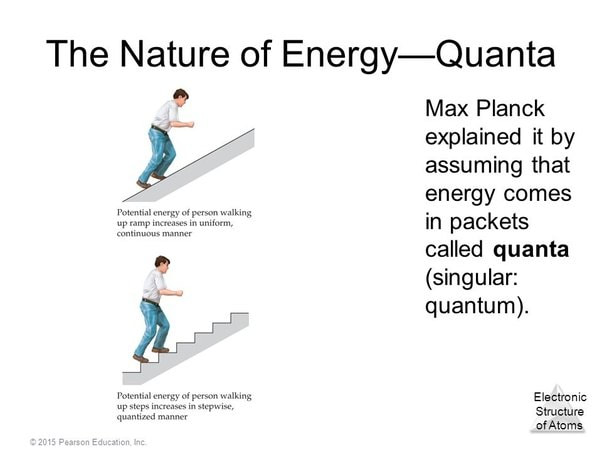
According to the theory quantum mechanics, an electron bound to an atom can not have any value of energy, rather it can only occupy certain states which correspond to certain energy levels. Because an electron bound to an atom can only have certain energies the electron can only absorb photons of certain energies exactly matched to the energy difference, or “quantum leap”, between two energy states.
In physics and chemistry, a quantum is a discrete packet of energy or matter. A photon is a single quantum of light. Light and other electromagnetic energy is absorbed or emitted in quanta or packets. A photon or the quantum of light belonging to the electromagnetic spectrum is released or absorbed during transition. When an electron absorbs a photon it gains the energy of the photon. Because an electron bound to an atom can only have certain energies the electron can only absorb photons of certain energies.
In physics and chemistry, a quantum is a discrete packet of energy or matter. A photon is a single quantum of light. Light and other electromagnetic energy is absorbed or emitted in quanta or packets. A photon or the quantum of light belonging to the electromagnetic spectrum is released or absorbed during transition. When an electron absorbs a photon it gains the energy of the photon. Because an electron bound to an atom can only have certain energies the electron can only absorb photons of certain energies.
eXCITED & GROUND STATE
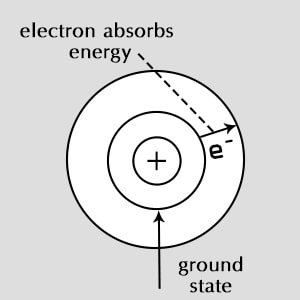
Ground State
✤ The state in which the electrons of an atom hold the lowest possible energies. Ground state can also be termed as the 'vacuum state'.
✤ The electrons belong to levels which do not have any energies linked with them. This level is termed as 'Zero energy level'.
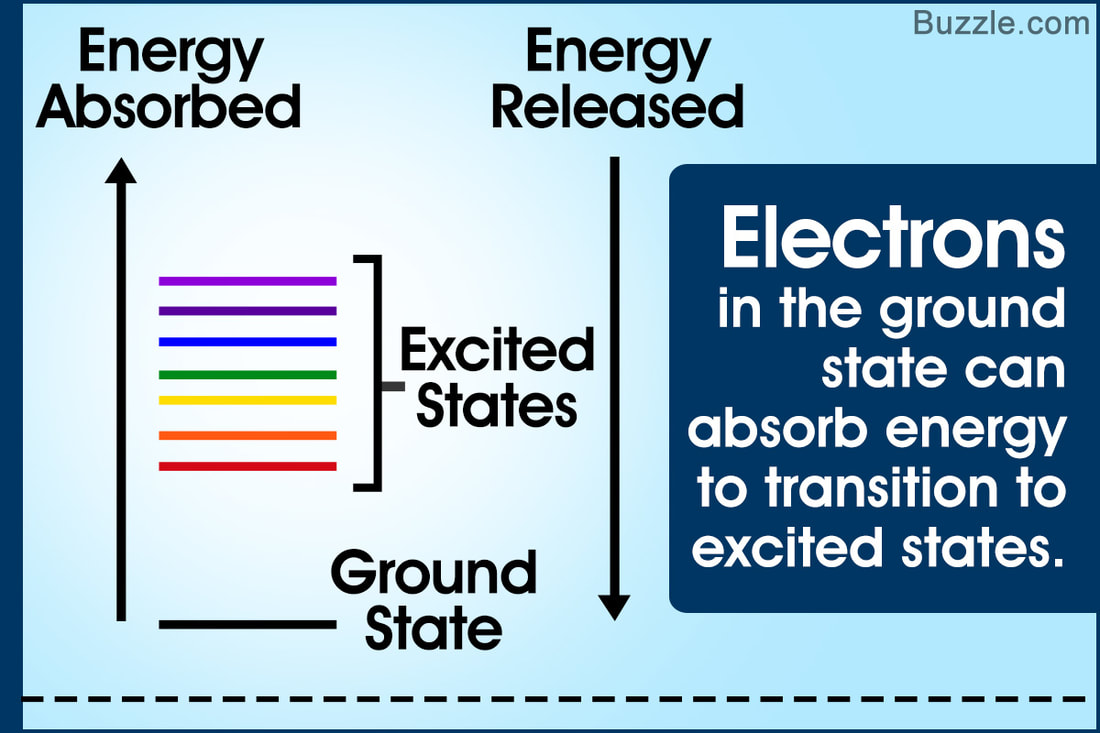
✤ The transition from ground to excited state is possible if electrons gain energy more than the difference in energies between the two energy orbits.
✤ The state in which the electrons of an atom hold the lowest possible energies. Ground state can also be termed as the 'vacuum state'.
✤ The electrons belong to levels which do not have any energies linked with them. This level is termed as 'Zero energy level'.
✤ The transition from ground to excited state is possible if electrons gain energy more than the difference in energies between the two energy orbits.
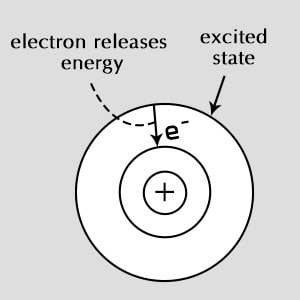
Excited State
✤ The state in which the electron energy levels are higher than those of the ground level. That is, not all the electrons will be in their lowest energy orbit.
✤ The excited electrons belong to levels which have energies associated with them.
✤ The transition from excited to ground state takes place when the electron drops down to its ground state by emitting a photon, which results in the display of an electromagnetic spectrum.
An electron is normally in its ground state, the lowest energy state available. After absorbing energy, it may jump from the ground state to a higher energy level, called an excited state.
✤ The state in which the electron energy levels are higher than those of the ground level. That is, not all the electrons will be in their lowest energy orbit.
✤ The excited electrons belong to levels which have energies associated with them.
✤ The transition from excited to ground state takes place when the electron drops down to its ground state by emitting a photon, which results in the display of an electromagnetic spectrum.
An electron is normally in its ground state, the lowest energy state available. After absorbing energy, it may jump from the ground state to a higher energy level, called an excited state.
atomic emission spectrum
The emission lines correspond to photons of discrete energies that are emitted when excited atomic states in the gas make transitions back to lower-lying levels.
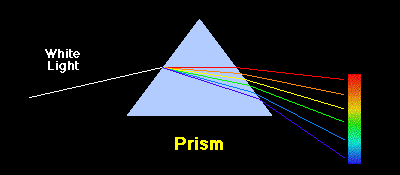
As we have noted in the section on the Bohr atom, isolated atoms can absorb and emit packets of electromagnetic radiation having discrete energies dictated by the detailed atomic structure of the atoms. When the corresponding light is passed through a prism or spectrograph it is separated spatially according to wavelength, as illustrated below.
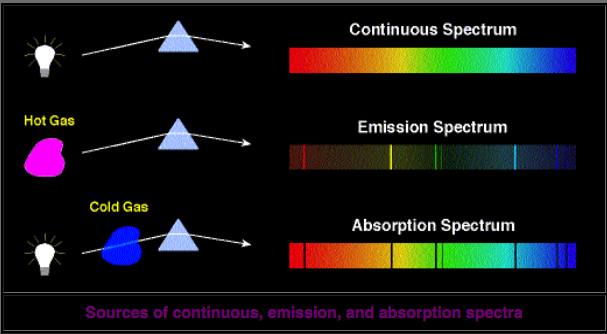
The corresponding spectrum may exhibit a continuum, or may have superposed on the continuum bright lines (an emission spectrum) or dark lines (an absorption spectrum), as illustrated in the following figure.
As we have noted in the section on the Bohr atom, isolated atoms can absorb and emit packets of electromagnetic radiation having discrete energies dictated by the detailed atomic structure of the atoms. When the corresponding light is passed through a prism or spectrograph it is separated spatially according to wavelength, as illustrated below.
The corresponding spectrum may exhibit a continuum, or may have superposed on the continuum bright lines (an emission spectrum) or dark lines (an absorption spectrum), as illustrated in the following figure.